|
This equation, derived from classical physics and using wave phenomena, infers that as wavelength increases, the intensity of energy provided will decrease with an inverse-squared relationship. 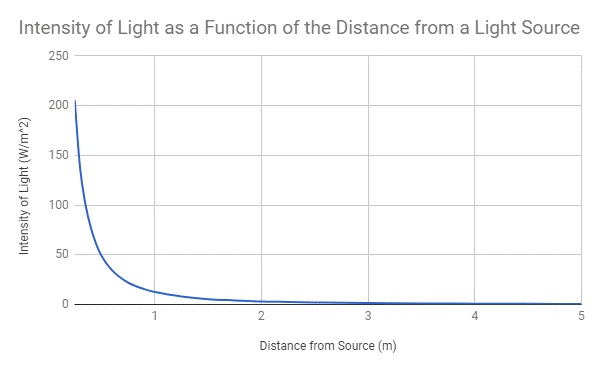
The relationship given by Planck's radiation law, given below, shows that with increasing temperature, the total radiated energy of a body increases and the peak of the emitted spectrum shifts to shorter wavelengths. The prevailing theory at the time of Max Planck’s discovery was that intensity and frequency were related by the equation I 2 k T 2. Even at very low light intensities, the photoelectric effect still occurs because the interaction is between one electron and one photon. While Planck originally regarded the hypothesis of dividing energy into increments as a mathematical artifice, introduced merely to get the correct answer, other physicists including Albert Einstein built on his work, and Planck's insight is now recognized to be of fundamental importance to quantum theory.Įvery physical body spontaneously and continuously emits electromagnetic radiation and the spectral radiance of a body, B ν, describes the spectral emissive power per unit area, per unit solid angle and per unit frequency for particular radiation frequencies. The light intensity corresponds to the number of photons arriving at the metal surface per unit time. The tables present conversion factors for light intensity and light sum for various types of light. In 1900, German physicist Max Planck heuristically derived a formula for the observed spectrum by assuming that a hypothetical electrically charged oscillator in a cavity that contained black-body radiation could only change its energy in a minimal increment, E, that was proportional to the frequency of its associated electromagnetic wave. One last remark is that the intensity of. Īt the end of the 19th century, physicists were unable to explain why the observed spectrum of black-body radiation, which by then had been accurately measured, diverged significantly at higher frequencies from that predicted by existing theories. In physics, Planck's law (also Planck radiation law : 1305 ) describes the spectral density of electromagnetic radiation emitted by a black body in thermal equilibrium at a given temperature T, when there is no net flow of matter or energy between the body and its environment. One formula for light intensity is I nfh At, I n f h A t, where: n n is the number of photons h h is Planck's constant f f is the frequency A A is the incident area t t is time. The classical (black) curve diverges from observed intensity at high frequencies (short wavelengths). The amount of energy passing through a unit area per unit time is the intensity of the wave. Shown here are a family of curves for different temperatures. Planck's law accurately describes black-body radiation. Not to be confused with Planck relation or Planck's principle.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
